Clinic Capabilities
This state-of-the-art clinical research facility is purpose-built to advance innovation in pharmaceuticals, dietary supplements, and medical devices. Designed with hospital-grade ventilation and a fully accessible infrastructure, it provides a controlled and high-performance environment for complex clinical studies.
With access to specialized laboratory services enabling precise and efficient biological sample analysis, SNI Clinics support every stage of research—from early product validation through to late-phase clinical trials—ensuring data integrity, participant safety, and scientifically robust outcomes across self-care and pharmaceutical sectors.
Our team also brings strong experience in generating and interpreting product-specific safety and efficacy data tailored to your formulation or technology. By aligning study design with your innovation and commercialization goals, we help substantiate claims, meet regulatory expectations, and accelerate your path to market.

End-to-End Clinical Research Expertise
Advance your innovation with customized clinical research solutions designed to generate strong, evidence-based results. Our experienced team integrates scientific insight with regulatory expertise to design and execute studies that align with your product’s specific goals and compliance requirements. From protocol development to post-market evaluation, we provide strategic guidance that enhances study efficiency, minimizes risk, and supports a faster path to commercialization.
Backed by over two decades of proven experience in both Canadian and U.S. regulatory environments, SNI delivers reliable, high-quality research that helps you validate safety, demonstrate efficacy, and achieve market success with confidence.

Facility and Equipment
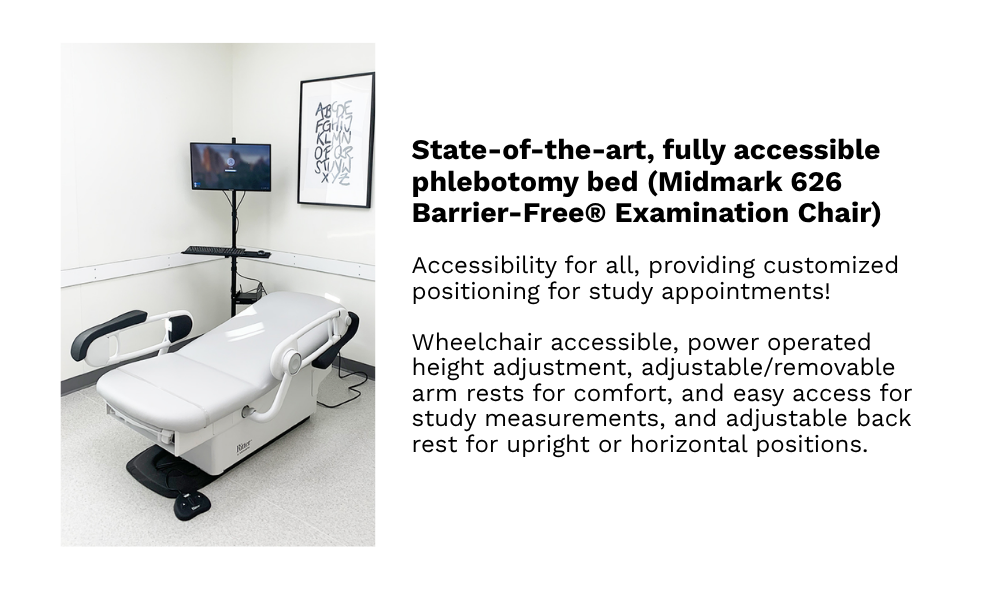
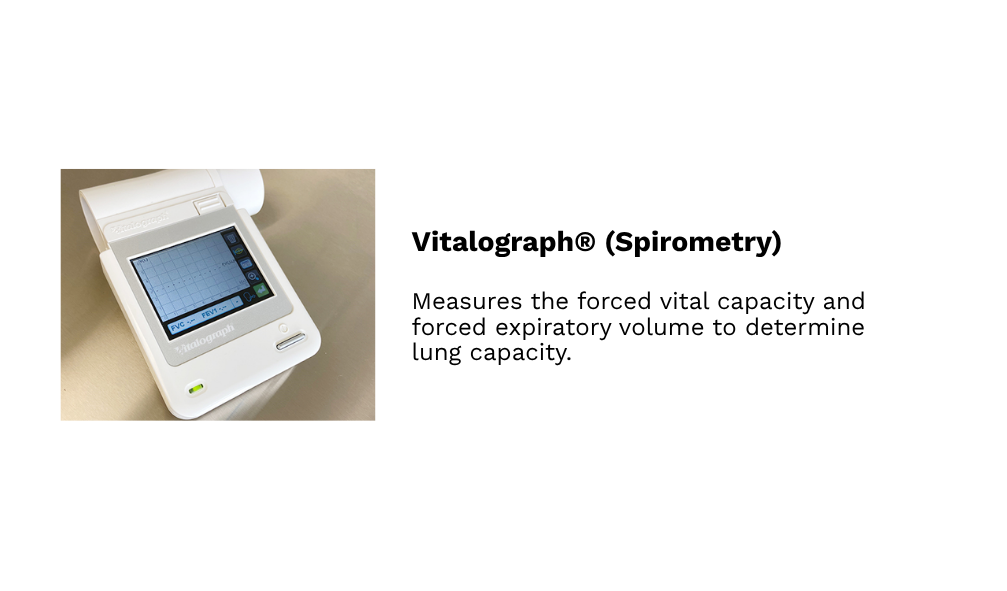
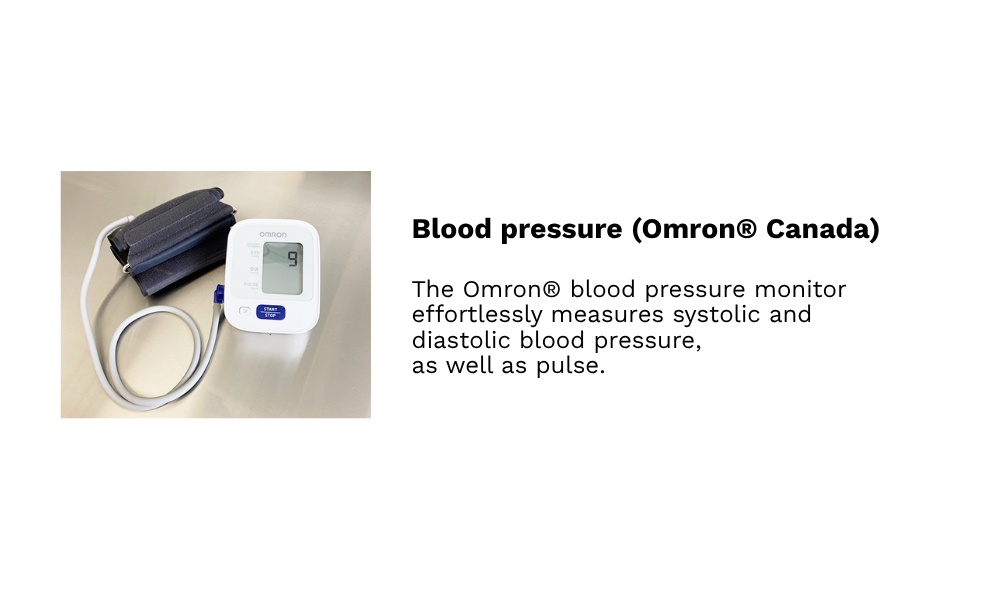
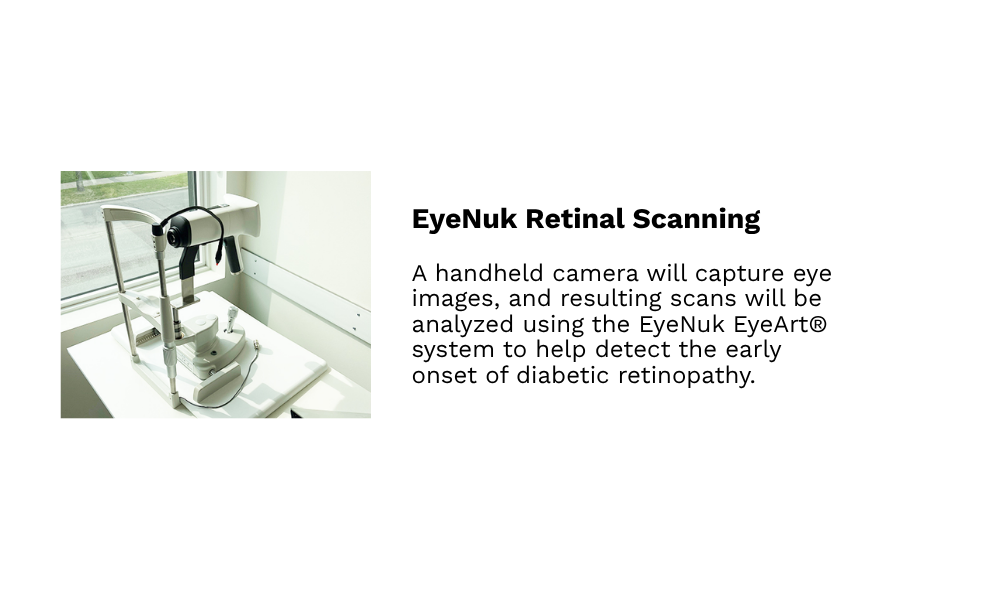
Our hospital-grade, custom-built, and fully accessible clinical research facilities in Winnipeg provide access to a diverse population of over 800,000 individuals, including participants from remote northern regions. Equipped with private examination rooms, on-site sample management, and advanced analytical systems, the facilities offer a controlled, professionally monitored environment that meets the highest standards of research quality.
Each space supports precise data collection, secure sample handling, and participant comfort, while advanced technologies and streamlined workflows ensure GCP compliance and deliver scientifically robust, reliable outcomes.
Ready to take your product to the next stage of development?
Book a consultation with our clinical research team to discuss your study objectives, product claims of interest, and evidence generation strategy. Our experts will guide you through every step, from protocol design to data analysis, ensuring your trial meets standards for safety, efficacy, and compliance.
Contact us today to talk to a member of our team!